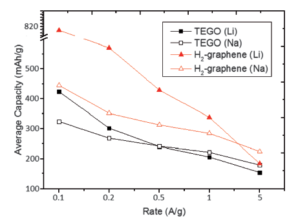
 The performance of graphene, and a few selected derivatives, was investigated as a negative electrode material in sodium and lithium-ion batteries. Hydrogenated graphene shows significant improvement in battery performance compared with as-prepared graphene, with reversible capacities of 488 mAh/g for lithium-ion batteries after 50 cycles and 491 mAh/g for sodium-ion batteries after 20 cycles. Notably, high rates of 1 A/g for graphene and 5 A/g for hydrogenated graphene indicate higher capacities in sodium-ion batteries than in lithium-ion batteries. Alternatively, nickel-nanoparticle decorated graphene performed relatively poorly in lithium-ion batteries. However, in sodium-ion batteries they showed the highest reversible capacities of all studied batteries and graphene derivatives, with 826 mAh/g after 25 cycles with 97% coulombic efficiency. Overall, minor modifications to graphene can dramatically improve electrochemical performance in both lithium-ion and sodium-ion batteries.
The performance of graphene, and a few selected derivatives, was investigated as a negative electrode material in sodium and lithium-ion batteries. Hydrogenated graphene shows significant improvement in battery performance compared with as-prepared graphene, with reversible capacities of 488 mAh/g for lithium-ion batteries after 50 cycles and 491 mAh/g for sodium-ion batteries after 20 cycles. Notably, high rates of 1 A/g for graphene and 5 A/g for hydrogenated graphene indicate higher capacities in sodium-ion batteries than in lithium-ion batteries. Alternatively, nickel-nanoparticle decorated graphene performed relatively poorly in lithium-ion batteries. However, in sodium-ion batteries they showed the highest reversible capacities of all studied batteries and graphene derivatives, with 826 mAh/g after 25 cycles with 97% coulombic efficiency. Overall, minor modifications to graphene can dramatically improve electrochemical performance in both lithium-ion and sodium-ion batteries.
Reproduced with permission. Copyright 2015, John Wiley & Sons
